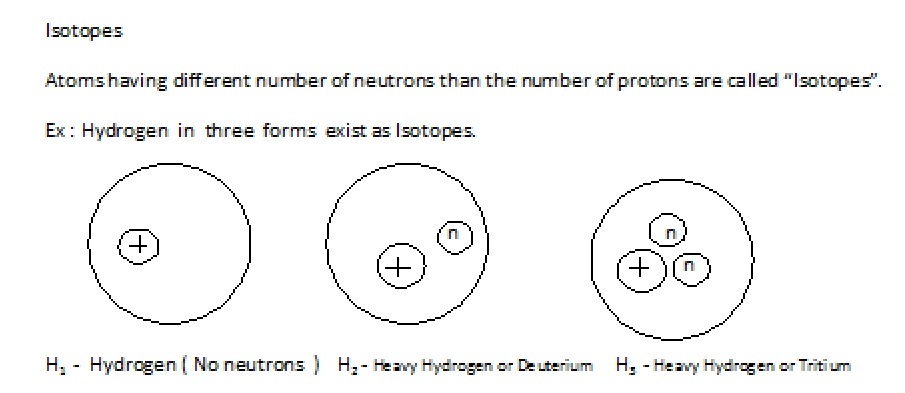
But in general in any atom the number of electrons = number of protons. This property is independent of neutron in the nucleus.
In periodic table occupy the same place since they have same chemical properties and same atomic number.
Radioactivity
Becquerel discovered the radioactivity in 1896. This phenomenon is confined in heaviest element in periodic table from 83 to 106 .
Spontaneous emission of powerful radiations exhibit by the heavy elements are called Radioactivity.
As elements changes its kind it is a drastic process activity of nuclear phenomenon.
Elements break itself . So it is irreversible self –disintegrating spontaneous activity Those elements exhibits this activity are called Radioactive elements.
Ex- Uranium, Polonium, radium, radon, ionium,thorium,actinium and mesothorium.
Radioactive elements emits the following radiations.
Alpha radiations α or alpha particles
Beta radiations β or beta particles
Gama radiation γ or photons
The natural elements that radiates these alpha , beta, gama rays are called Naturral Radioactivity.
If such rays are induced from some light elements artificially with some modern technique of transmutation of elements they are called Artificial Radioactivity.
In many other element much lighter than those that occur in nature such type of Radioactivity is known as artificial or induced radioactivity.
The five type of radiation ( with electrical charges indicated as + - for positive and negative respectively and the mass in atomic mass units ) are
Gamma rays or photons - electromagnetic radiation.
Neutrons uncharged particles approximate mass = 1
Photons + 1 charged particles, mass approximately = 1
Alpha particles helium nuclei charge +2 mass 4
Beta particles electrons charge = 1, positrons ( charge +1 ), mass very small.
Binding Energy
The nucleus of an atom is formed when the nucleons come closer to each other and the distance between two nucleons is of the order of nearly 10-12 mm. At the moment of combination there is a release of energy and is known as Binding Energy.
In periodic table occupy the same place since they have same chemical properties and same atomic number.
Radioactivity
Becquerel discovered the radioactivity in 1896. This phenomenon is confined in heaviest element in periodic table from 83 to 106 .
Spontaneous emission of powerful radiations exhibit by the heavy elements are called Radioactivity.
As elements changes its kind it is a drastic process activity of nuclear phenomenon.
Elements break itself . So it is irreversible self –disintegrating spontaneous activity Those elements exhibits this activity are called Radioactive elements.
Ex- Uranium, Polonium, radium, radon, ionium,thorium,actinium and mesothorium.
Radioactive elements emits the following radiations.
Alpha radiations α or alpha particles
Beta radiations β or beta particles
Gama radiation γ or photons
The natural elements that radiates these alpha , beta, gama rays are called Naturral Radioactivity.
If such rays are induced from some light elements artificially with some modern technique of transmutation of elements they are called Artificial Radioactivity.
In many other element much lighter than those that occur in nature such type of Radioactivity is known as artificial or induced radioactivity.
The five type of radiation ( with electrical charges indicated as + - for positive and negative respectively and the mass in atomic mass units ) are
Gamma rays or photons - electromagnetic radiation.
Neutrons uncharged particles approximate mass = 1
Photons + 1 charged particles, mass approximately = 1
Alpha particles helium nuclei charge +2 mass 4
Beta particles electrons charge = 1, positrons ( charge +1 ), mass very small.
Binding Energy
The nucleus of an atom is formed when the nucleons come closer to each other and the distance between two nucleons is of the order of nearly 10-12 mm. At the moment of combination there is a release of energy and is known as Binding Energy.